Construction of Relief System for Adverse Reactions of Traditional Chinese Medicine Injections in China
-
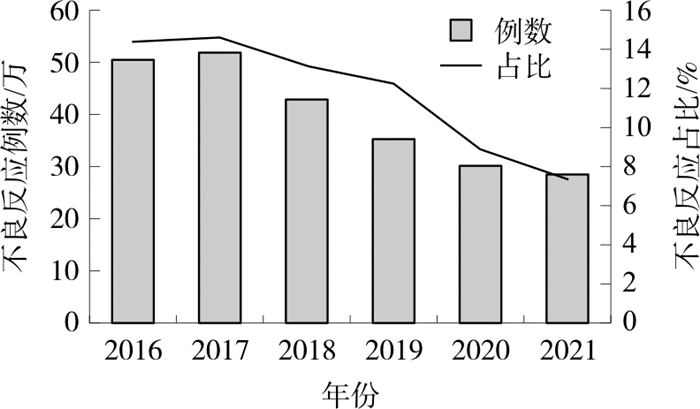
摘要: 中药注射剂是我国自主研制的突破传统给药方式的新剂型, 在现代医疗实践中发挥了至关重要的作用,然而,中药注射剂不良反应事件的频发引起了人们广泛关注。因中药注射剂不良反应事件导致患者严重损害的, 相关主体应对受害人进行赔偿。但基于缺陷认定存在争议、归责原则仍未明确、救济机制尚未建立的现实困境,现行的法律救济在实践操作中尚有欠缺,无法对受害者提供有效救济。通过借鉴国外药品不良反应救济制度的成功经验,结合我国具体国情, 建议完善药品缺陷解释标准,明确中药注射剂不良反应中的责任归属,设立专项补偿基金,强化对基金环节的治理与监督,建立以基金模式为主的中药注射剂不良反应的补偿制度。Abstract: The traditional Chinese medicine (TCM) injection is a new dosage form independently developed in China, which breaks through the traditional way of administration and plays a vital role in modern medical practice. However, the frequent occurrences of adverse reactions of TCM injections have aroused widespread concern. If the adverse reaction events of TCM injection cause serious damage to the patient, the relevant subject shall compensate the victims. However, based on the realistic dilemma such as controversial defect identification, undefined principle of liability fixation, and unestablished relief mechanism has not yet been established, there is defectiveness of the current legal relief in practical operation and it cannot provide effective relief to victims. By drawing on the successful experience of foreign adverse reaction relief systems combined with China's specific national conditions, it is suggested to improve the interpretation standards of drug defects, clarify the responsibility attribution in the adverse reactions of TCM injections, establish a special compensation fund, strengthen the governance and supervision of the fund link, as well as establish a compensation system for the adverse reactions of TCM injections mainly based on the fund model.
-
Key words:
- traditional Chinese medicine injection /
- adverse reaction /
- relief system
-
[1] 何自会, 王青, 刘沫, 等. 中药注射剂质量控制研究进展[J]. 中国医药工业杂志, 2021, 52(5): 599-610. https://www.cnki.com.cn/Article/CJFDTOTAL-ZHOU202105003.htm [2] 国家食品药品监督管理总局. 国家药品不良反应监测年度报告(2021年)[EB/OL]. [2022-03-30]. https://www.nmpa.gov.cn/directory/web/nmpa/xxgk/yjjsh/ypblfytb/20220329161925106.html. [3] 彭国平, 李存玉. 中药注射剂安全性的分析与思考[J]. 南京中医药大学学报, 2019, 35(6): 744-751. https://www.cnki.com.cn/Article/CJFDTOTAL-NJZY201906026.htm [4] 田侃, 赵文敏, 杨毅. 再论中药注射剂的合理性——基于红花注射液和喜炎平注射液召回事件的思考[J]. 中国药房, 2018, 29(18): 2449-2452. https://www.cnki.com.cn/Article/CJFDTOTAL-ZGYA201818001.htm [5] 王珂欣, 高丽, 秦雪梅, 等. 中药注射剂色差与安全性研究进展[J]. 中草药, 2019, 50(9): 2219-2223. https://www.cnki.com.cn/Article/CJFDTOTAL-ZCYO201909030.htm [6] 谭乐俊, 王萌, 朱彦. 中药注射剂的不良反应研究进展[J]. 中国中药杂志, 2014, 39(20): 3889-3898. https://www.cnki.com.cn/Article/CJFDTOTAL-ZGZY201420002.htm [7] 张文晋, 曹也, 张燕, 等. 中药注射剂原料中药材实施GAP现状及展望[J]. 中国中药杂志, 2021, 46(20): 5166-5172. https://www.cnki.com.cn/Article/CJFDTOTAL-ZGZY202120002.htm [8] 贺红强. 论我国药品不良反应的归责原则[J]. 中国卫生事业管理, 2010, 27(12): 827-831. https://www.cnki.com.cn/Article/CJFDTOTAL-ZWSG201012014.htm [9] 邹宜諠, 陈云, 柳鹏程, 等. 德国和日本药品上市许可持有人制度下的药品损害事件风险管理研究[J]. 中国药事, 2018, 32(3): 362-366. https://www.cnki.com.cn/Article/CJFDTOTAL-ZGYS201803012.htm [10] 刘智媛. 药品不良反应损害救济制度探究[D]. 济南: 山东大学, 2014. [11] 陈云, 任磊, 颜建周, 等. MAH制度下日本药品不良反应损害救济基金制度研究[J]. 中国药事, 2017, 31(12): 1395-1403. https://www.cnki.com.cn/Article/CJFDTOTAL-ZGYS201712007.htm [12] MAEDA H, KUROKAWA T. Regulatory review time for approval of oncology drugs in Japan between 2001 and 2014. Considerations of changes, factors that affect review time, and difference with the United States[J]. J Clin Pharmacol, 2015, 55(5): 481-489. [13] 王慧, 赵丽, 徐巍. 发达国家及我国台湾地区药品不良反应损害救济制度的介绍及思考[J]. 药物流行病学杂志, 2018, 27(2): 138-141. https://www.cnki.com.cn/Article/CJFDTOTAL-YWLX201802015.htm [14] 张枫, 徐晓媛. 日本与我国台湾地区药品不良反应损害救济基金制度的比较及对我国的启示[J]. 中国药房, 2017, 28(22): 3036-3039. https://www.cnki.com.cn/Article/CJFDTOTAL-ZGYA201722004.htm [15] 沈群红, 张诗情, 崔诗月. 药品不良反应赔偿制度的国际比较与借鉴[J]. 中国卫生政策研究, 2016, 9(9): 41-46. https://www.cnki.com.cn/Article/CJFDTOTAL-ZGWZ201609008.htm [16] 李新, 刘胤岐, 臧恒昌. 国外药物不良反应补偿模式对我国补偿模式的启示[J]. 中国继续医学教育, 2019, 11(34): 88-91. https://www.cnki.com.cn/Article/CJFDTOTAL-JXUY201934036.htm [17] 华东. 我国药品不良反应救济制度的建立[J]. 中医药管理杂志, 2014, 22(4): 492-493. https://www.cnki.com.cn/Article/CJFDTOTAL-ZYYG201404005.htm -




 下载:
下载: